When President Donald Trump signed an executive order in December to expedite marijuana rescheduling, he emphasized opening up research on therapeutic uses of cannabinoids, particularly CBD for the elderly.
In fact, the order itself is titled, “Increasing Medical Marijuana and Cannabidiol Research.”
Flanked by Health and Human Services Secretary Robert F. Kennedy Jr. and other researchers and doctors, Trump said, “This reclassification order will make it far easier to conduct medical marijuana-related research, allowing us to study benefits, potential dangers and future treatments.”
Kennedy added that because of a lack of federal studies, much of the available evidence about the effectiveness, dangers and proper dosage of cannabinoids is anecdotal and hypothetical. “This will finally allow us to study this issue and answer these questions for the American people,” he said.
But like legalization, banking and interstate commerce, reclassifying marijuana from a Schedule I to a Schedule III controlled substance may not live up to the hype for research, experts say.
Delayed cannabis regulation, approvals
More than three months since Trump’s Dec. 18 order, the Department of Justice has still not posted a rule to reclassify marijuana on the Federal Register. The rescheduling process, which began in 2024 under President Joe Biden, has been held up through procedural hearings.
Chief Administrative Law Judge John Mulrooney, who was hearing the matter, retired on Aug. 1, 2025. He noted that the Controlled Substances Act requires an administrative law judge to oversee proceedings under the Administrative Procedure Act.
“My retirement will leave the DEA with no Administrative Law Judge to hear this matter or any of the Agency’s other pending administrative actions,” he said in his retirement notice.
Rep. Steve Cohen, D-Tenn., asked Attorney General Pam Bondi and DEA Administrator Terrance Cole about the expected timeline to complete rulemaking in a March 27 letter. He said he believes marijuana should never have been placed in Schedule I.
“Rescheduling is widely understood to be in alignment with the best available scientific data and will significantly strengthen scientific and medical research into marijuana,” he wrote.
Since then, Trump fired Bondi and appointed Deputy Attorney General Todd Blanche as acting attorney general. He is reportedly considering Environmental Protection Agency Administrator Lee Zeldin to be the permanent replacement. Zeldin has had a mixed record on legalizing cannabis, according to Marijuana Moment, so it’s unclear how quickly rescheduling will recommence.
But even if cannabis is ultimately moved to Schedule III, research will be limited by Federal Drug Administration and Drug Enforcement Administration approvals, a 2022 law co-written by Smart Approaches to Marijuana (SAM), and ultimately, funding.
Marijuana research is currently possible, but it requires FDA approval of an Investigational New Drug application. And the plant material must be obtained from a DEA-registered research cultivator. Currently, there are seven registered cultivators, but not all of them are producing cannabis.
According to the Cannabis Regulators Association (CANNRA), obtaining registration as a Schedule III facility might be easier, but it won’t necessarily change the availability of cannabis material.
The University of Mississippi’s National Center for Natural Products Research is the longest holder of a DEA cultivation license. Don Stanford, assistant director of the university’s Research Institute of Pharmaceutical Sciences, believes Schedule III will advance clinical research and product development efforts.
“For instance, an interventional study that requires the clinical site to handle and store test articles will be able to proceed under a DEA C-III registration, which is commonplace in clinics and pharmacies. Additionally, C-III clinical research materials will become more widely available and less expensive for researchers,” Stanford wrote in a statement to CRB Monitor News.
He said the university’s Resource Center for Cannabis and Cannabinoid Research will be rolling out a materials exchange program soon.
“The new regulations for acquisition and handling of C-III materials will streamline utilization of this ‘peer-to-peer’ network that will enable collaboration between DEA-licensed researchers,” said Stanford.
However, the restrictions of Schedule III research are still onerous and costly, according to Dr. Sue Sisley, president of DEA-registered Scottsdale Research Institute in Arizona. For example, she says the storage change simply means she would be able to keep cannabis in an expensive cage rather than in a safe.
“As long as it’s managed by the FDA, there will be relentless barriers,” she said.
Meanwhile, the Medical Marijuana and Cannabidiol Research Expansion Act (MCREA) of 2022 places more restrictions on cannabis, regardless of where it sits on the schedule. Touted as a way to increase cannabis research at the time, prohibition group SAM proudly helped write the legislation.
“The historic signing of the Medical Marijuana and Cannabidiol Research Expansion Act demonstrates that we can lower barriers to marijuana research without descheduling and legalizing marijuana,” SAM President Kevin Sabet said in Dec. 2, 2022, press release. “Unlike bills like the SAFE Banking Act, MORE Act, and the Cannabis Administration & Opportunity Act, this legislation represents needed and meaningful reform to marijuana research.”
But industry insiders said MCREA actually makes cannabis research more complicated. Meanwhile, research licensing rules under MCREA have not been promulgated by the DEA, according to CANNRA.
“It remains unclear if the Schedule III research requirements can and will be used by DEA given the passage of MCREA,” CANNRA said in a December 2025 fact sheet on the implications of rescheduling. “While less restrictive than Schedule I requirements, research requirements under MCREA are more restrictive than those under Schedule III.”
MCREA also requires the attorney general and secretary of Health and Human Services to write annual reports on the adequacy of uninterrupted supply. Within one year, HHS and the National Institutes of Health (NIH) were to submit a report to congressional committees on the “potential therapeutic effects of cannabidiol or marijuana on serious medical conditions,” and other effects of delta-9 THC, as well as barriers to research.
During an HHS budget hearing in March 2024, Sen. Elizabeth Warren, D-Mass., asked when should members anticipate release of the report. HHS responded, “The report will be submitted to Congress as soon as it is completed.”
An HHS spokesperson did not respond by deadline to a question asking if the report was ever written.
Cannabis research costs money
At the end of the day, the availability of funding will determine whether there will be more research.
Dr. Peter Grinspoon, a board-certified addiction specialist at Massachusetts General Hospital, noted that Trump’s order was “exciting” and had a lot of symbolic meaning because the medical industry urgently needs more clinical research. Most of the studies available have been anecdotal, not randomized, controlled studies.
“Tons of what we do in medicine is observational evidence,” he said, including in pregnant women and children. “Why are tens of millions of Americans using it if it doesn’t work?”
In his practice, he’s found success using cannabis to help people recover from opioid and alcohol addiction as part of harm reduction. But for those with cannabis addiction, there is no FDA-approved medication to treat it.
Grinspoon said we may get real-world research, but pharmaceutical companies are not interested in funding it.
“Who’s going to pay for these randomized controlled trials?” he asked.
Sisley agrees. She said Trump’s order would be better if it came with a “moonshot” of funding.
“There’s no demand for research cannabis because there is no funding. How can there be a surge if nobody is paying for it?”
MCREA states, “To the extent practicable, the Secretary of Health and Human Services, either directly or through awarding grants, contacts, or cooperative agreements, shall expand and coordinate the activities of the National Institutes of Health and other relevant Federal agencies to better determine the effects of cannabidiol and marijuana.”
From fiscal year 2022 to 2024, NIH received $657 million for cannabinoid research through grants, contracts and other funding mechanisms. It had another $143 million for cannabidiol research. Funding for fiscal years 2025 and 2026 are not available “due to evolving changes in administration priorities,” the NIH said on its website.
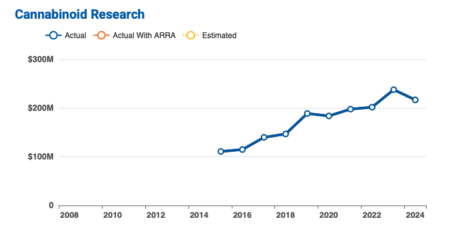
Dr. Nora Volkow, director of the NIH’s National Institute on Drug Abuse, joined Trump at the signing and said Schedule III placement would accelerate their research work.
According to a NIH Record newsletter on Jan. 16, the NIH invested $21.7 million in cannabinoid research in 2024, although it had $217 million in funding. One study, the Adolescent Brain Cognitive Development Study, is following 10,000 young people from adolescence to young adulthood to inform on prevention interventions.
CBD and hemp show more progress
Another focus of Trump’s order was using CBD for pain treatment, particularly for seniors. CBD can also be found in hemp, not just marijuana. More progress is being made in making CBD more accessible for seniors as the Centers for Medicare & Medicaid Services announced a program where some seniors can be reimbursed up to $500 for CBD products. A rule was filed in the Federal Register on April 6.
“This is the first government-led testing of quality and outcomes of patients across different conditions. And it delivers on the need for more data collection and research into hemp usage. CMS is going to collect data,” CMS Administrator Dr. Mehmet Oz said about the Innovation Center programs during the signing ceremony.
The policy changes fly in the face of a looming ban in November on CBD products with more than 0.4 milligrams of THC, which industry leaders say will practically shut down the $28 billion hemp market.
Grinspoon, who is also an instructor at Harvard Medical School, has written a book to educate seniors about using CBD, “Aging Well with Cannabis: Feel Better, Sleep Better and Live Better with Marijuana and CBD.”
He said the policy juxtaposition of recriminalizing hemp while the government is willing to pay for it is “incoherent.” He said making it illegal will only push people toward the illicit market.
Patients in states where marijuana is still illegal will suffer the most, he said. “There is no legal marijuana market to be rerouting them through.”













